In Which Process Do Animals Turn Food Energy
Every bit we have just seen, cells require a abiding supply of energy to generate and maintain the biological club that keeps them alive. This free energy is derived from the chemical bail energy in food molecules, which thereby serve as fuel for cells.
Sugars are peculiarly important fuel molecules, and they are oxidized in small steps to carbon dioxide (COtwo) and h2o (Figure 2-69). In this section we trace the major steps in the breakdown, or catabolism, of sugars and evidence how they produce ATP, NADH, and other activated carrier molecules in fauna cells. We concentrate on glucose breakup, since it dominates energy product in most animal cells. A very similar pathway also operates in plants, fungi, and many bacteria. Other molecules, such as fat acids and proteins, tin can also serve every bit energy sources when they are funneled through appropriate enzymatic pathways.

Figure 2-69
Schematic representation of the controlled stepwise oxidation of sugar in a cell, compared with ordinary called-for. (A) In the cell, enzymes catalyze oxidation via a series of small steps in which free free energy is transferred in conveniently sized packets (more...)
Food Molecules Are Broken Down in Three Stages to Produce ATP
The proteins, lipids, and polysaccharides that make upwards most of the food we swallow must exist cleaved down into smaller molecules before our cells can utilize them—either equally a source of energy or as building blocks for other molecules. The breakdown processes must act on food taken in from outside, but non on the macromolecules within our own cells. Stage 1 in the enzymatic breakdown of food molecules is therefore digestion, which occurs either in our intestine outside cells, or in a specialized organelle within cells, the lysosome. (A membrane that surrounds the lysosome keeps its digestive enzymes separated from the cytosol, as described in Chapter 13.) In either case, the large polymeric molecules in food are broken down during digestion into their monomer subunits—proteins into amino acids, polysaccharides into sugars, and fats into fat acids and glycerol—through the action of enzymes. Subsequently digestion, the small organic molecules derived from food enter the cytosol of the prison cell, where their gradual oxidation begins. Equally illustrated in Effigy 2-70, oxidation occurs in two further stages of cellular catabolism: stage 2 starts in the cytosol and ends in the major free energy-converting organelle, the mitochondrion; stage 3 is entirely confined to the mitochondrion.
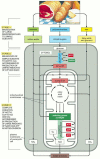
Figure 2-seventy
Simplified diagram of the three stages of cellular metabolism that lead from food to waste products in animal cells. This serial of reactions produces ATP, which is then used to bulldoze biosynthetic reactions and other energy-requiring processes in the (more...)
In stage 2 a chain of reactions chosen glycolysis converts each molecule of glucose into 2 smaller molecules of pyruvate. Sugars other than glucose are similarly converted to pyruvate after their conversion to 1 of the sugar intermediates in this glycolytic pathway. During pyruvate formation, two types of activated carrier molecules are produced—ATP and NADH. The pyruvate so passes from the cytosol into mitochondria. There, each pyruvate molecule is converted into COii plus a two-carbon acetyl group—which becomes attached to coenzyme A (CoA), forming acetyl CoA, another activated carrier molecule (see Effigy 2-62). Large amounts of acetyl CoA are as well produced by the stepwise breakdown and oxidation of fatty acids derived from fats, which are carried in the bloodstream, imported into cells equally fatty acids, and then moved into mitochondria for acetyl CoA product.
Phase 3 of the oxidative breakdown of food molecules takes place entirely in mitochondria. The acetyl group in acetyl CoA is linked to coenzyme A through a high-energy linkage, and it is therefore easily transferable to other molecules. Afterwards its transfer to the four-carbon molecule oxaloacetate, the acetyl group enters a series of reactions called the citric acid wheel. As nosotros hash out presently, the acetyl grouping is oxidized to COii in these reactions, and big amounts of the electron carrier NADH are generated. Finally, the loftier-energy electrons from NADH are passed along an electron-transport chain within the mitochondrial inner membrane, where the energy released by their transfer is used to drive a process that produces ATP and consumes molecular oxygen (O2). It is in these concluding steps that most of the energy released by oxidation is harnessed to produce most of the cell'south ATP.
Because the energy to drive ATP synthesis in mitochondria ultimately derives from the oxidative breakdown of food molecules, the phosphorylation of ADP to form ATP that is driven by electron transport in the mitochondrion is known every bit oxidative phosphorylation. The fascinating events that occur within the mitochondrial inner membrane during oxidative phosphorylation are the major focus of Chapter 14.
Through the production of ATP, the free energy derived from the breakup of sugars and fats is redistributed equally packets of chemical energy in a form convenient for use elsewhere in the prison cell. Roughly 109 molecules of ATP are in solution in a typical prison cell at any instant, and in many cells, all this ATP is turned over (that is, used up and replaced) every 1–2 minutes.
In all, about half of the free energy that could in theory exist derived from the oxidation of glucose or fatty acids to H2O and CO2 is captured and used to bulldoze the energetically unfavorable reaction Pi + ADP → ATP. (By contrast, a typical combustion engine, such as a car engine, can convert no more than 20% of the bachelor free energy in its fuel into useful work.) The residue of the energy is released by the cell as heat, making our bodies warm.
Glycolysis Is a Key ATP-producing Pathway
The most important process in stage 2 of the breakdown of nutrient molecules is the degradation of glucose in the sequence of reactions known equally glycolysis—from the Greek glukus, "sweet," and lusis, "rupture." Glycolysis produces ATP without the interest of molecular oxygen (O2 gas). Information technology occurs in the cytosol of most cells, including many anaerobic microorganisms (those that can live without utilizing molecular oxygen). Glycolysis probably evolved early in the history of life, before the activities of photosynthetic organisms introduced oxygen into the atmosphere. During glycolysis, a glucose molecule with six carbon atoms is converted into ii molecules of pyruvate, each of which contains iii carbon atoms. For each molecule of glucose, two molecules of ATP are hydrolyzed to provide energy to drive the early on steps, but four molecules of ATP are produced in the subsequently steps. At the terminate of glycolysis, at that place is consequently a net gain of two molecules of ATP for each glucose molecule broken downward.
The glycolytic pathway is presented in outline in Figure 2-71, and in more than detail in Console 2-viii (pp. 124–125). Glycolysis involves a sequence of 10 separate reactions, each producing a different sugar intermediate and each catalyzed by a unlike enzyme. Similar most enzymes, these enzymes all take names ending in ase—like isomerase and dehydrogenase—which point the type of reaction they catalyze.
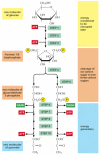
Figure 2-71
An outline of glycolysis. Each of the 10 steps shown is catalyzed by a different enzyme. Note that step four cleaves a six-carbon sugar into two 3-carbon sugars, so that the number of molecules at every phase later on this doubles. As indicated, step half-dozen (more than...)
![]()
Panel 2-8
Details of the ten Steps of Glycolysis.
Although no molecular oxygen is involved in glycolysis, oxidation occurs, in that electrons are removed by NAD+ (producing NADH) from some of the carbons derived from the glucose molecule. The stepwise nature of the process allows the energy of oxidation to be released in small packets, and then that much of information technology can exist stored in activated carrier molecules rather than all of it being released as rut (see Figure 2-69). Thus, some of the energy released by oxidation drives the straight synthesis of ATP molecules from ADP and Pi, and some remains with the electrons in the high-energy electron carrier NADH.
2 molecules of NADH are formed per molecule of glucose in the form of glycolysis. In aerobic organisms (those that crave molecular oxygen to live), these NADH molecules donate their electrons to the electron-send chain described in Chapter 14, and the NAD+ formed from the NADH is used again for glycolysis (see step 6 in Console 2-eight, pp. 124–125).
Fermentations Let ATP to Be Produced in the Absence of Oxygen
For virtually beast and plant cells, glycolysis is only a prelude to the third and terminal phase of the breakdown of food molecules. In these cells, the pyruvate formed at the last step of stage 2 is rapidly transported into the mitochondria, where information technology is converted into CO2 plus acetyl CoA, which is and then completely oxidized to CO2 and H2O.
In contrast, for many anaerobic organisms—which practise not utilize molecular oxygen and can abound and divide without it—glycolysis is the primary source of the prison cell's ATP. This is also true for certain beast tissues, such as skeletal muscle, that tin can proceed to office when molecular oxygen is limiting. In these anaerobic conditions, the pyruvate and the NADH electrons stay in the cytosol. The pyruvate is converted into products excreted from the cell—for case, into ethanol and CO2 in the yeasts used in brewing and breadmaking, or into lactate in muscle. In this process, the NADH gives upward its electrons and is converted back into NAD+. This regeneration of NAD+ is required to maintain the reactions of glycolysis (Effigy 2-72).
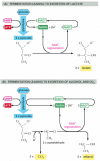
Effigy 2-72
Two pathways for the anaerobic breakdown of pyruvate. (A) When inadequate oxygen is present, for example, in a muscle cell undergoing vigorous contraction, the pyruvate produced by glycolysis is converted to lactate as shown. This reaction regenerates (more...)
Anaerobic energy-yielding pathways like these are called fermentations. Studies of the commercially important fermentations carried out by yeasts inspired much of early on biochemistry. Work in the nineteenth century led in 1896 to the and then startling recognition that these processes could be studied outside living organisms, in prison cell extracts. This revolutionary discovery eventually fabricated it possible to dissect out and study each of the individual reactions in the fermentation procedure. The piecing together of the complete glycolytic pathway in the 1930s was a major triumph of biochemistry, and it was apace followed by the recognition of the primal part of ATP in cellular processes. Thus, virtually of the key concepts discussed in this chapter accept been understood for more than 50 years.
Glycolysis Illustrates How Enzymes Couple Oxidation to Energy Storage
We take previously used a "paddle bike" analogy to explain how cells harvest useful energy from the oxidation of organic molecules by using enzymes to couple an energetically unfavorable reaction to an energetically favorable one (see Effigy 2-56). Enzymes play the part of the paddle cycle in our illustration, and we at present return to a step in glycolysis that nosotros accept previously discussed, in lodge to illustrate exactly how coupled reactions occur.
2 key reactions in glycolysis (steps 6 and 7) convert the three-carbon saccharide intermediate glyceraldehyde 3-phosphate (an aldehyde) into 3-phosphoglycerate (a carboxylic acid). This entails the oxidation of an aldehyde group to a carboxylic acid grouping, which occurs in 2 steps. The overall reaction releases enough free energy to convert a molecule of ADP to ATP and to transfer 2 electrons from the aldehyde to NAD+ to form NADH, while still releasing enough heat to the environment to make the overall reaction energetically favorable (ΔG° for the overall reaction is -3.0 kcal/mole).
The pathway by which this remarkable feat is accomplished is outlined in Effigy two-73. The chemical reactions are guided by two enzymes to which the sugar intermediates are tightly bound. The beginning enzyme (glyceraldehyde 3-phosphate dehydrogenase) forms a short-lived covalent bail to the aldehyde through a reactive -SH group on the enzyme, and it catalyzes the oxidation of this aldehyde while yet in the attached state. The high-energy enzyme-substrate bond created past the oxidation is then displaced past an inorganic phosphate ion to produce a high-energy sugar-phosphate intermediate, which is thereby released from the enzyme. This intermediate so binds to the second enzyme (phosphoglycerate kinase). This enzyme catalyzes the energetically favorable transfer of the high-energy phosphate just created to ADP, forming ATP and completing the process of oxidizing an aldehyde to a carboxylic acrid (meet Figure 2-73).

Effigy 2-73
Energy storage in steps 6 and vii of glycolysis. In these steps the oxidation of an aldehyde to a carboxylic acid is coupled to the formation of ATP and NADH. (A) Footstep vi begins with the germination of a covalent bond between the substrate (glyceraldehyde (more than...)
We have shown this particular oxidation process in some detail because it provides a clear case of enzyme-mediated energy storage through coupled reactions (Figure 2-74). These reactions (steps 6 and seven) are the merely ones in glycolysis that create a high-free energy phosphate linkage straight from inorganic phosphate. As such, they business relationship for the cyberspace yield of two ATP molecules and two NADH molecules per molecule of glucose (run into Panel 2-8, pp. 124–125).

Figure 2-74
Schematic view of the coupled reactions that form NADH and ATP in steps vi and 7 of glycolysis. The C-H bond oxidation free energy drives the formation of both NADH and a high-energy phosphate bond. The breakage of the high-free energy bond then drives ATP germination. (more...)
Every bit we take just seen, ATP can exist formed readily from ADP when reaction intermediates are formed with higher-energy phosphate bonds than those in ATP. Phosphate bonds can exist ordered in energy by comparing the standard complimentary-energy change (ΔYard°) for the breakage of each bail past hydrolysis. Figure 2-75 compares the high-free energy phosphoanhydride bonds in ATP with other phosphate bonds, several of which are generated during glycolysis.

Effigy 2-75
Some phosphate bail energies. The transfer of a phosphate group from whatsoever molecule 1 to any molecule ii is energetically favorable if the standard complimentary-energy change (ΔG°) for the hydrolysis of the phosphate bond in molecule 1 is more than negative (more...)
Sugars and Fats Are Both Degraded to Acetyl CoA in Mitochondria
We now motion on to consider stage iii of catabolism, a process that requires abundant molecular oxygen (Oii gas). Since the Earth is thought to have developed an temper containing O2 gas betwixt one and two billion years ago, whereas abundant life-forms are known to have existed on the Globe for three.v billion years, the use of O2 in the reactions that we talk over side by side is thought to be of relatively recent origin. In contrast, the mechanism used to produce ATP in Figure ii-73 does not require oxygen, and relatives of this elegant pair of coupled reactions could have arisen very early in the history of life on Globe.
In aerobic metabolism, the pyruvate produced by glycolysis is rapidly decarboxylated by a behemothic complex of three enzymes, called the pyruvate dehydrogenase complex. The products of pyruvate decarboxylation are a molecule of COtwo (a waste product), a molecule of NADH, and acetyl CoA. The three-enzyme complex is located in the mitochondria of eucaryotic cells; its structure and mode of action are outlined in Figure 2-76.

Figure two-76
The oxidation of pyruvate to acetyl CoA and CO2. (A) The construction of the pyruvate dehydrogenase complex, which contains 60 polypeptide chains. This is an instance of a big multienzyme complex in which reaction intermediates are passed directly from (more...)
The enzymes that degrade the fatty acids derived from fats as well produce acetyl CoA in mitochondria. Each molecule of fatty acid (as the activated molecule fatty acyl CoA) is broken downward completely past a bike of reactions that trims two carbons at a fourth dimension from its carboxyl terminate, generating 1 molecule of acetyl CoA for each turn of the cycle. A molecule of NADH and a molecule of FADHtwo are also produced in this process (Figure 2-77).

Effigy 2-77
The oxidation of fatty acids to acetyl CoA. (A) Electron micrograph of a lipid droplet in the cytoplasm (top), and the structure of fats (bottom). Fats are triacylglycerols. The glycerol portion, to which 3 fatty acids are linked through ester bonds, (more than...)
Sugars and fats provide the major energy sources for near non-photosynthetic organisms, including humans. However, the majority of the useful energy that can be extracted from the oxidation of both types of foodstuffs remains stored in the acetyl CoA molecules that are produced by the 2 types of reactions just described. The citric acrid cycle of reactions, in which the acetyl group in acetyl CoA is oxidized to CO2 and HiiO, is therefore central to the free energy metabolism of aerobic organisms. In eucaryotes these reactions all take place in mitochondria, the organelle to which pyruvate and fat acids are directed for acetyl CoA production (Figure two-78). We should therefore not be surprised to notice that the mitochondrion is the place where about of the ATP is produced in creature cells. In contrast, aerobic leaner conduct out all of their reactions in a single compartment, the cytosol, and it is here that the citric acrid cycle takes identify in these cells.

Figure two-78
Pathways for the production of acetyl CoA from sugars and fats. The mitochondrion in eucaryotic cells is the place where acetyl CoA is produced from both types of major food molecules. It is therefore the place where most of the cell'southward oxidation reactions (more...)
The Citric Acid Cycle Generates NADH by Oxidizing Acetyl Groups to CO2
In the nineteenth century, biologists noticed that in the absence of air (anaerobic conditions) cells produce lactic acrid (for example, in muscle) or ethanol (for example, in yeast), while in its presence (aerobic weather) they consume Otwo and produce COii and H2O. Intensive efforts to define the pathways of aerobic metabolism eventually focused on the oxidation of pyruvate and led in 1937 to the discovery of the citric acid wheel, also known as the tricarboxylic acid cycle or the Krebs cycle. The citric acid cycle accounts for nigh two-thirds of the total oxidation of carbon compounds in most cells, and its major end products are CO2 and high-energy electrons in the form of NADH. The COii is released as a waste product, while the high-energy electrons from NADH are passed to a membrane-spring electron-transport chain, eventually combining with O2 to produce H2O. Although the citric acid cycle itself does non use O2, it requires O2 in gild to proceed because at that place is no other efficient mode for the NADH to get rid of its electrons and thus regenerate the NAD+ that is needed to keep the wheel going.
The citric acid bicycle, which takes place inside mitochondria in eucaryotic cells, results in the complete oxidation of the carbon atoms of the acetyl groups in acetyl CoA, converting them into CO2. But the acetyl group is not oxidized directly. Instead, this group is transferred from acetyl CoA to a larger, four-carbon molecule, oxaloacetate, to class the six-carbon tricarboxylic acid, citric acid, for which the subsequent cycle of reactions is named. The citric acid molecule is then gradually oxidized, assuasive the free energy of this oxidation to be harnessed to produce energy-rich activated carrier molecules. The chain of eight reactions forms a wheel because at the end the oxaloacetate is regenerated and enters a new turn of the cycle, every bit shown in outline in Effigy 2-79.

Effigy two-79
Simple overview of the citric acid cycle. The reaction of acetyl CoA with oxaloacetate starts the wheel by producing citrate (citric acrid). In each turn of the cycle, two molecules of CO2 are produced as waste products, plus three molecules of NADH, 1 (more...)
We accept thus far discussed only one of the 3 types of activated carrier molecules that are produced by the citric acid cycle, the NAD+-NADH pair (see Figure ii-60). In addition to three molecules of NADH, each turn of the bicycle besides produces ane molecule of FADH 2 (reduced flavin adenine dinucleotide) from FAD and ane molecule of the ribonucleotide GTP (guanosine triphosphate) from GDP. The structures of these ii activated carrier molecules are illustrated in Figure 2-80. GTP is a shut relative of ATP, and the transfer of its concluding phosphate grouping to ADP produces one ATP molecule in each wheel. Like NADH, FADH2 is a carrier of high-energy electrons and hydrogen. Every bit we talk over soon, the energy that is stored in the readily transferred high-energy electrons of NADH and FADH2 will be utilized later on for ATP production through the process of oxidative phosphorylation, the but step in the oxidative catabolism of foodstuffs that direct requires gaseous oxygen (O2) from the atmosphere.
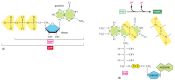
Figure two-fourscore
The structures of GTP and FADH2. (A) GTP and Gdp are close relatives of ATP and ADP, respectively. (B) FADH2 is a carrier of hydrogens and high-energy electrons, like NADH and NADPH. It is shown here in its oxidized form (FAD) with the hydrogen-carrying (more...)
The complete citric acrid bike is presented in Panel 2-ix (pp. 126–127). The actress oxygen atoms required to make COtwo from the acetyl groups entering the citric acid cycle are supplied not by molecular oxygen, simply by h2o. As illustrated in the panel, three molecules of water are split up in each cycle, and the oxygen atoms of some of them are ultimately used to brand COii.
![]()
In improver to pyruvate and fat acids, some amino acids pass from the cytosol into mitochondria, where they are also converted into acetyl CoA or 1 of the other intermediates of the citric acid cycle. Thus, in the eucaryotic cell, the mitochondrion is the center toward which all free energy-yielding processes lead, whether they begin with sugars, fats, or proteins.
The citric acid cycle as well functions as a starting indicate for important biosynthetic reactions by producing vital carbon-containing intermediates, such as oxaloacetate and α-ketoglutarate. Some of these substances produced past catabolism are transferred back from the mitochondrion to the cytosol, where they serve in anabolic reactions as precursors for the synthesis of many essential molecules, such equally amino acids.
Electron Transport Drives the Synthesis of the Bulk of the ATP in Most Cells
Information technology is in the last footstep in the degradation of a food molecule that the major portion of its chemic energy is released. In this final process the electron carriers NADH and FADH2 transfer the electrons that they have gained when oxidizing other molecules to the electron-transport chain, which is embedded in the inner membrane of the mitochondrion. As the electrons pass along this long chain of specialized electron acceptor and donor molecules, they autumn to successively lower energy states. The energy that the electrons release in this process is used to pump H+ ions (protons) across the membrane—from the inner mitochondrial compartment to the outside (Figure two-81). A slope of H+ ions is thereby generated. This slope serves equally a source of free energy, being tapped like a bombardment to drive a variety of energy-requiring reactions. The virtually prominent of these reactions is the generation of ATP by the phosphorylation of ADP.

Effigy two-81
The generation of an H+ slope beyond a membrane by electron-ship reactions. A high-energy electron (derived, for example, from the oxidation of a metabolite) is passed sequentially by carriers A, B, and C to a lower energy state. In this diagram (more...)
At the end of this serial of electron transfers, the electrons are passed to molecules of oxygen gas (Oii) that have diffused into the mitochondrion, which simultaneously combine with protons (H+) from the surrounding solution to produce molecules of h2o. The electrons have now reached their lowest energy level, and therefore all the bachelor energy has been extracted from the food molecule being oxidized. This procedure, termed oxidative phosphorylation (Figure 2-82), also occurs in the plasma membrane of bacteria. Every bit one of the virtually remarkable achievements of cellular evolution, information technology volition be a central topic of Chapter 14.
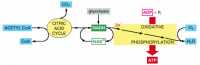
Effigy 2-82
The final stages of oxidation of food molecules. Molecules of NADH and FADH2 (FADH2 is not shown) are produced by the citric acid wheel. These activated carriers donate loftier-free energy electrons that are somewhen used to reduce oxygen gas to water. A major (more than...)
In total, the complete oxidation of a molecule of glucose to H2O and CO2 is used by the cell to produce about xxx molecules of ATP. In contrast, just 2 molecules of ATP are produced per molecule of glucose by glycolysis lone.
Organisms Shop Food Molecules in Special Reservoirs
All organisms need to maintain a high ATP/ADP ratio, if biological order is to be maintained in their cells. Withal animals have simply periodic access to nutrient, and plants demand to survive overnight without sunlight, without the possibility of sugar product from photosynthesis. For this reason, both plants and animals convert sugars and fats to special forms for storage (Effigy ii-83).

Figure two-83
The storage of sugars and fats in creature and plant cells. (A) The structures of starch and glycogen, the storage class of sugars in plants and animals, respectively. Both are storage polymers of the saccharide glucose and differ merely in the frequency of branch (more than...)
To compensate for long periods of fasting, animals shop fatty acids as fat aerosol equanimous of h2o-insoluble triacylglycerols, largely in specialized fat cells. And for shorter-term storage, saccharide is stored as glucose subunits in the large branched polysaccharide glycogen, which is present as pocket-sized granules in the cytoplasm of many cells, including liver and musculus. The synthesis and degradation of glycogen are rapidly regulated co-ordinate to need. When more than ATP is needed than can be generated from the food molecules taken in from the bloodstream, cells suspension down glycogen in a reaction that produces glucose i-phosphate, which enters glycolysis.
Quantitatively, fatty is a far more important storage form than glycogen, in role because the oxidation of a gram of fat releases about twice as much free energy every bit the oxidation of a gram of glycogen. Moreover, glycogen differs from fat in binding a dandy deal of water, producing a sixfold difference in the actual mass of glycogen required to store the same amount of energy as fat. An boilerplate adult man stores enough glycogen for only about a day of normal activities but plenty fat to last for nearly a calendar month. If our principal fuel reservoir had to exist carried as glycogen instead of fat, trunk weight would need to be increased past an average of virtually lx pounds.
Most of our fat is stored in adipose tissue, from which it is released into the bloodstream for other cells to utilize as needed. The need arises afterwards a menstruum of not eating; even a normal overnight fast results in the mobilization of fat, so that in the forenoon most of the acetyl CoA entering the citric acid wheel is derived from fatty acids rather than from glucose. After a meal, yet, most of the acetyl CoA entering the citric acid cycle comes from glucose derived from food, and whatever excess glucose is used to replenish depleted glycogen stores or to synthesize fats. (While animal cells readily convert sugars to fats, they cannot catechumen fatty acids to sugars.)
Although plants produce NADPH and ATP past photosynthesis, this important process occurs in a specialized organelle, chosen a chloroplast, which is isolated from the remainder of the plant cell by a membrane that is impermeable to both types of activated carrier molecules. Moreover, the found contains many other cells—such as those in the roots—that lack chloroplasts and therefore cannot produce their own sugars or ATP. Therefore, for most of its ATP product, the plant relies on an export of sugars from its chloroplasts to the mitochondria that are located in all cells of the plant. Most of the ATP needed by the plant is synthesized in these mitochondria and exported from them to the residuum of the plant cell, using exactly the aforementioned pathways for the oxidative breakup of sugars that are utilized past nonphotosynthetic organisms (Figure 2-84).

Figure 2-84
How the ATP needed for most plant cell metabolism is made. In plants, the chloroplasts and mitochondria collaborate to supply cells with metabolites and ATP.
During periods of excess photosynthetic capacity during the day, chloroplasts convert some of the sugars that they brand into fats and into starch, a polymer of glucose analogous to the glycogen of animals. The fats in plants are triacylglycerols, simply like the fats in animals, and differ merely in the types of fatty acids that predominate. Fat and starch are both stored in the chloroplast as reservoirs to be mobilized as an energy source during periods of darkness (see Figure 2-83B).
The embryos within establish seeds must alive on stored sources of free energy for a prolonged period, until they germinate to produce leaves that tin harvest the energy in sunlight. For this reason plant seeds ofttimes contain specially large amounts of fats and starch—which makes them a major food source for animals, including ourselves (Figure ii-85).

Figure 2-85
Some found seeds that serve as important foods for humans. Corn, basics, and peas all contain rich stores of starch and fatty that provide the young plant embryo in the seed with free energy and building blocks for biosynthesis. (Courtesy of the John Innes Foundation.) (more...)
Amino Acids and Nucleotides Are Part of the Nitrogen Cycle
In our discussion so far we take full-bodied mainly on carbohydrate metabolism. We have non notwithstanding considered the metabolism of nitrogen or sulfur. These two elements are constituents of proteins and nucleic acids, which are the two nigh important classes of macromolecules in the jail cell and make up approximately 2-thirds of its dry weight. Atoms of nitrogen and sulfur pass from chemical compound to compound and between organisms and their environment in a serial of reversible cycles.
Although molecular nitrogen is arable in the Earth's atmosphere, nitrogen is chemically unreactive equally a gas. Just a few living species are able to incorporate information technology into organic molecules, a procedure called nitrogen fixation. Nitrogen fixation occurs in sure microorganisms and by some geophysical processes, such equally lightning discharge. It is essential to the biosphere equally a whole, for without it life would not exist on this planet. Merely a minor fraction of the nitrogenous compounds in today'due south organisms, however, is due to fresh products of nitrogen fixation from the atmosphere. Most organic nitrogen has been in circulation for some time, passing from one living organism to another. Thus nowadays-day nitrogen-fixing reactions tin be said to perform a "topping-up" office for the total nitrogen supply.
Vertebrates receive almost all of their nitrogen in their dietary intake of proteins and nucleic acids. In the body these macromolecules are broken down to amino acids and the components of nucleotides, and the nitrogen they contain is used to produce new proteins and nucleic acids or utilized to make other molecules. Most one-half of the xx amino acids found in proteins are essential amino acids for vertebrates (Figure ii-86), which means that they cannot exist synthesized from other ingredients of the diet. The others tin can exist so synthesized, using a variety of raw materials, including intermediates of the citric acid cycle every bit described beneath. The essential amino acids are made by nonvertebrate organisms, unremarkably by long and energetically expensive pathways that take been lost in the form of vertebrate development.

Figure 2-86
The nine essential amino acids. These cannot be synthesized by homo cells and then must be supplied in the diet.
The nucleotides needed to make RNA and Dna tin can be synthesized using specialized biosynthetic pathways: there are no "essential nucleotides" that must be provided in the diet. All of the nitrogens in the purine and pyrimidine bases (equally well as some of the carbons) are derived from the plentiful amino acids glutamine, aspartic acid, and glycine, whereas the ribose and deoxyribose sugars are derived from glucose.
Amino acids that are not utilized in biosynthesis tin can be oxidized to generate metabolic energy. Nigh of their carbon and hydrogen atoms eventually form COii or HiiO, whereas their nitrogen atoms are shuttled through various forms and eventually appear equally urea, which is excreted. Each amino acid is processed differently, and a whole constellation of enzymatic reactions exists for their catabolism.
Many Biosynthetic Pathways Begin with Glycolysis or the Citric Acid Cycle
Catabolism produces both energy for the cell and the building blocks from which many other molecules of the cell are made (see Figure 2-36). Thus far, our discussions of glycolysis and the citric acrid cycle have emphasized free energy production, rather than the provision of the starting materials for biosynthesis. But many of the intermediates formed in these reaction pathways are also siphoned off past other enzymes that use them to produce the amino acids, nucleotides, lipids, and other small organic molecules that the cell needs. Some idea of the complexity of this procedure tin be gathered from Figure two-87, which illustrates some of the branches from the primal catabolic reactions that atomic number 82 to biosyntheses.
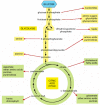
Figure 2-87
Glycolysis and the citric acid bicycle provide the precursors needed to synthesize many important biological molecules. The amino acids, nucleotides, lipids, sugars, and other molecules—shown here as products—in plough serve as the precursors (more...)
The existence of so many branching pathways in the jail cell requires that the choices at each co-operative exist carefully regulated, equally we discuss next.
Metabolism Is Organized and Regulated
One gets a sense of the intricacy of a jail cell equally a chemical machine from the relation of glycolysis and the citric acrid bike to the other metabolic pathways sketched out in Figure two-88. This type of chart, which was used earlier in this affiliate to innovate metabolism, represents simply some of the enzymatic pathways in a cell. It is obvious that our word of jail cell metabolism has dealt with just a tiny fraction of cellular chemistry.

Figure 2-88
Glycolysis and the citric acid bicycle are at the eye of metabolism. Some 500 metabolic reactions of a typical cell are shown schematically with the reactions of glycolysis and the citric acid bicycle in carmine. Other reactions either lead into these 2 (more...)
All these reactions occur in a jail cell that is less than 0.i mm in diameter, and each requires a different enzyme. As is clear from Figure 2-88, the same molecule tin often exist role of many different pathways. Pyruvate, for example, is a substrate for half a dozen or more than different enzymes, each of which modifies it chemically in a different way. 1 enzyme converts pyruvate to acetyl CoA, another to oxaloacetate; a third enzyme changes pyruvate to the amino acrid alanine, a quaternary to lactate, and and then on. All of these unlike pathways compete for the same pyruvate molecule, and similar competitions for thousands of other small molecules become on at the aforementioned time. A improve sense of this complexity can possibly be attained from a three-dimensional metabolic map that allows the connections between pathways to be made more direct (Figure two-89).
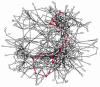
Figure 2-89
A representation of all of the known metabolic reactions involving small molecules in a yeast cell. Every bit in Figure 2-88, the reactions of glycolysis and the citric acid cycle are highlighted in red. This metabolic map is unusual in making utilize of three-dimensions, (more...)
The situation is further complicated in a multicellular organism. Different cell types volition in full general require somewhat different sets of enzymes. And different tissues make distinct contributions to the chemical science of the organism every bit a whole. In addition to differences in specialized products such as hormones or antibodies, in that location are pregnant differences in the "common" metabolic pathways among diverse types of cells in the same organism.
Although about all cells contain the enzymes of glycolysis, the citric acid bicycle, lipid synthesis and breakup, and amino acrid metabolism, the levels of these processes required in different tissues are not the same. For example, nervus cells, which are probably the most fastidious cells in the body, maintain almost no reserves of glycogen or fat acids and rely nearly entirely on a abiding supply of glucose from the bloodstream. In dissimilarity, liver cells supply glucose to actively contracting muscle cells and recycle the lactic acrid produced by muscle cells back into glucose (Figure two-90). All types of cells have their distinctive metabolic traits, and they cooperate extensively in the normal state, every bit well as in response to stress and starvation. One might recollect that the whole organization would need to be so finely counterbalanced that any small-scale upset, such as a temporary change in dietary intake, would exist disastrous.
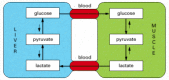
Figure 2-90
Schematic view of the metabolic cooperation between liver and musculus cells. The chief fuel of actively contracting muscle cells is glucose, much of which is supplied by liver cells. Lactic acid, the end product of anaerobic glucose breakup by glycolysis (more than...)
In fact, the metabolic balance of a cell is amazingly stable. Whenever the rest is perturbed, the jail cell reacts so equally to restore the initial land. The cell can conform and continue to role during starvation or disease. Mutations of many kinds can damage or even eliminate item reaction pathways, and even so—provided that sure minimum requirements are met—the cell survives. It does so considering an elaborate network of control mechanisms regulates and coordinates the rates of all of its reactions. These controls rest, ultimately, on the remarkable abilities of proteins to change their shape and their chemical science in response to changes in their immediate surround. The principles that underlie how large molecules such as proteins are built and the chemical science backside their regulation volition exist our side by side concern.
Summary
Glucose and other nutrient molecules are broken down by controlled stepwise oxidation to provide chemical energy in the form of ATP and NADH. These are three main sets of reactions that act in series—the products of each being the starting material for the side by side: glycolysis (which occurs in the cytosol), the citric acid cycle (in the mitochondrial matrix), and oxidative phosphorylation (on the inner mitochondrial membrane). The intermediate products of glycolysis and the citric acrid cycle are used both as sources of metabolic energy and to produce many of the small molecules used equally the raw materials for biosynthesis. Cells store carbohydrate molecules as glycogen in animals and starch in plants; both plants and animals as well employ fats extensively as a food store. These storage materials in turn serve equally a major source of food for humans, forth with the proteins that comprise the majority of the dry out mass of the cells we eat.
Source: https://www.ncbi.nlm.nih.gov/books/NBK26882/
Posted by: ybanezdiestlyped1957.blogspot.com

0 Response to "In Which Process Do Animals Turn Food Energy"
Post a Comment